A Murine Model of Contact Lens-associated Fusarium Keratitis
Abstract
Purpose:
Fusarium solani and F. oxysporum were the causative organisms of the 2005/2006 outbreak of contact lens-associated fungal keratitis in the United States. The present study was an investigation of the ability of F. oxysporum grown as a biofilm on silicone hydrogel contact lenses to induce keratitis.
Methods:
A clinical isolate of F. oxysporum was grown as a biofilm on lotrafilcon A contact lenses, and a 2-mm diameter punch was placed on the abraded corneal epithelium of either untreated or cyclophosphamide-treated C57BL/6 mice or of IL-1R1(-/-), MyD88(-/-), TLR2(-/-), or TLR4(-/-) mice. After 2 hours, the lens was removed, and corneal opacification, colony forming units (CFUs), and histopathology were evaluated.
Results:
C57BL/6 mice developed severe corneal opacification within 24 hours and resolved after four days. In contrast, corneal opacification progressed in cyclophosphamide-treated mice, and was associated with unimpaired fungal growth in the cornea, and with hyphae penetrating into the anterior chamber. The phenotype of MyD88(-/-) and IL-1R(-/-) mice was similar to that of cyclophosphamide-treated animals, with significantly impaired cellular infiltration and fungal clearance. Although TLR4(-/-) mice developed a cellular infiltrate and corneal opacification similar to C57BL/6 mice, the CFU count was significantly and consistently higher.
Conclusions:
Fusarium grown as a biofilm on silicone hydrogel contact lenses can induce keratitis on injured corneas, with disease severity and fungal killing dependent on the innate immune response, including IL-1R1, MyD88, and TLR4.
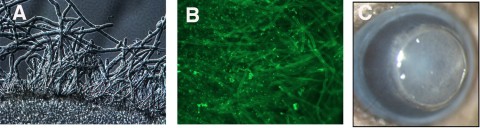
Figure 1
Murine model of Fusarium biofilm keratitis. Fusarium conidia were incubated 90 minutes with lotrafilcon A contact lenses, washed, and incubated a further 48 hours. (A) Fusarium hyphae associated with the contact lens (DIC microscopy). (B) After incubation with FITC-ConA, showing hyphae and extracellular matrix. (C) Murine model: 2-mm-diameter punch of a contact lens with attached Fusarium biofilm on the corneal surface of a C57BL/6 mouse. Original magnification: (A, B) ×400; (C) ×20.
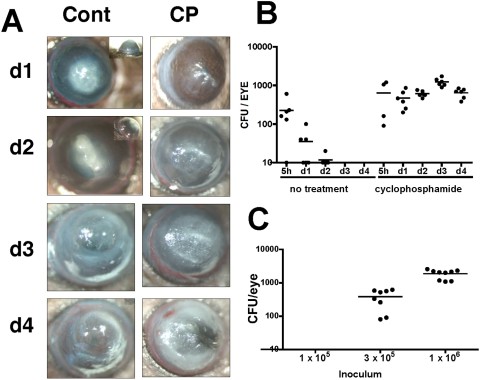
Figure 2
Biofilm-associated Fusarium keratitis in untreated and cyclophosphamide-treated C57BL/6 mice. C57BL/6 mice were either untreated or immunosuppressed by intraperitoneal injection of cyclophosphamide (CP). The corneas were abraded, and silicone hydrogel contact lenses (lotrafilcon) with Fusarium biofilm were placed on the ocular surface for 2 hours and examined at indicated time points thereafter. (A) Representative corneas of immunocompetent C57BL/6 mice, which were given a 1-mm-diameter abrasion, exhibited severe opacification on day 1 and day 2, and resolved after day 4. In contrast, corneas of CP-treated mice, which received three parallel scratches, showed increasing opacity, over time and perforation after day 4 (not shown). (B) Eyes of immunocompetent and immunosuppressed (CP) mice were enucleated on days 1 to 4, and CFUs were counted. Data points represent individual corneas and mice, and the data are combined from two repeat experiments. P < 0.05 between untreated and CP-treated mice at each time point except 5 hours. (C) The limit of detection of Fusarium colonies in CP-treated mice. Biofilm was generated with a starting inoculum as shown (1 × 105, 3 × 105, and 1 × 106 conidia per mL PBS). Corneas were then abraded and incubated with contact lenses with adherent biofilm, mice were euthanatized and CFUs were counted on day 1.
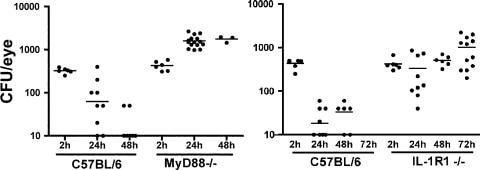
Figure 3
Fusarium survival in MyD88−/− and IL-1R1−/− mice. Corneas of C57BL/6, MyD88−/−, and IL-1R1−/− mice were abraded with three parallel scratches and exposed to contact lenses with adherent Fusarium biofilm. At each time point, the eyes were homogenized and the CFUs were assessed. Data points represent individual corneas and mice combined from two similar experiments. P < 0.05 between C57BL/6 and MyD88−/− mice at 24 and 48 hours, and between C57BL/6 and IL-1R1−/− mice at 24, 48, and 72 hours.
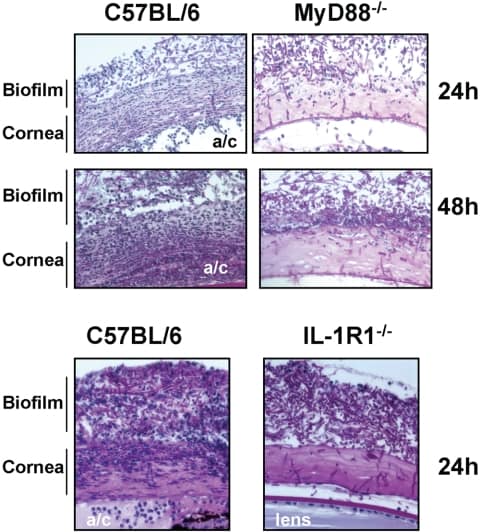
Figure 4
Histopathology of Fusarium keratitis in MyD88−/− and IL-1R1−/− mice. C57BL/6, MyD88−/−, and IL-1R1−/− mice were given a 1-mm diameter corneal abrasion and a contact lens with adherent Fusarium biofilm was placed on the corneal surface, as described in the legend to Figure 1. After 24 or 48 hours, the eyes were processed for histology and stained with PAS. Representative images of five mice per group are shown. Note that cellular infiltration of the corneal stroma was impaired in the MyD88−/− and IL-1R1−/− mice compared with that in the C57BL/6 mice. a/c, anterior chamber.
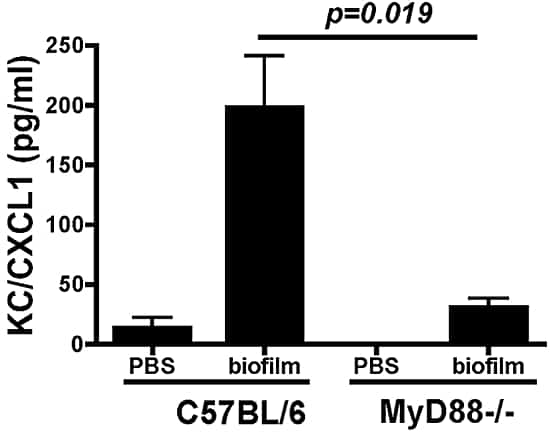
Figure 5
CXCL1/KC production in MyD88−/− mice after infection with Fusarium biofilm on contact lenses. The corneas of the C57BL/6 and MyD88−/− mice were abraded with three parallel scratches and exposed to lenses with adherent Fusarium biofilm. After 3 hours, the corneas were dissected and homogenized, and CXCL1/KC was measured by ELISA. Data are expressed as the mean ± SEM of five mice per group and are representative of results in two experiments.
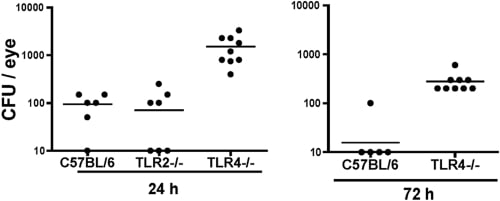
Figure 6
Fusarium keratitis in TLR2−/− and TLR4−/− mice. Corneas of C57BL/6, TLR2−/−, and TLR4−/− mice were abraded with three parallel scratches and exposed to contact lenses with Fusarium biofilm, as described in the legend to Figure 2. At 24 and 72 hours, the eyes were homogenized and CFUs were assessed. Data points represent individual corneas in two separate experiments. P < 0.001; ANOVA at 24 hours between C57BL/6 and TLR4−/− mice and between the TLR2−/− and TLR4−/− mice and 72-hour data between the C57BL/6 and TLR4−/− mice.